CAR-T is moving upstream — and Darzalex-era benchmarks are starting to look structurally capped.
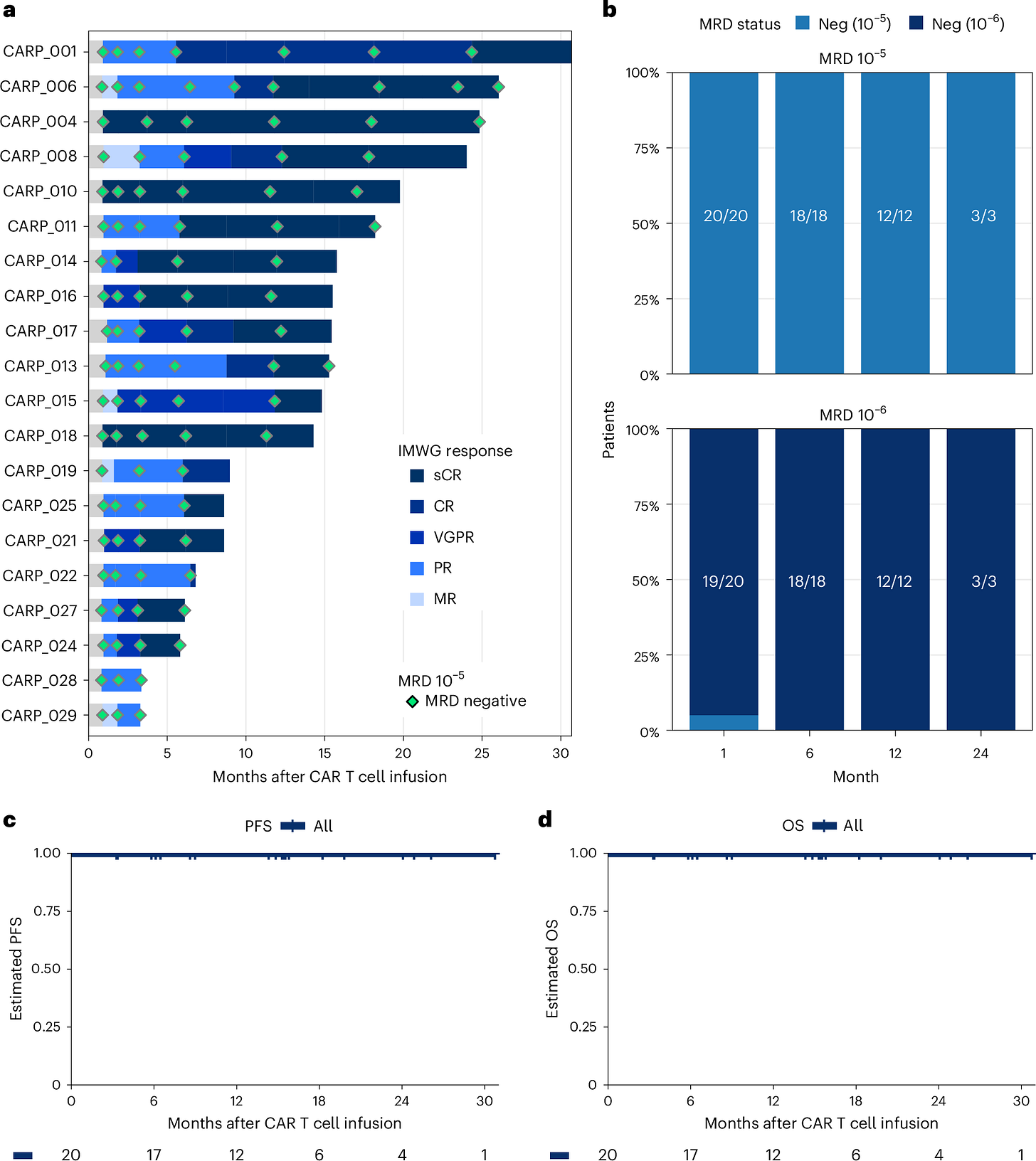
New phase 2 data from the Dana-Farber study “Phase 2 Trial Shows Deep and Lasting Responses to CAR T-Cell Therapy for High-Risk Smoldering Multiple Myeloma” evaluated Carvykti in high-risk smoldering multiple myeloma — a pre-myeloma setting where the disease is not yet symptomatic, but biologically primed for progression.
At 15.3 months median follow-up:
100% MRD negativity (10⁻⁶) across all treated patients
100% overall response rate
~90% complete or stringent complete responses
No disease progression or deaths observed
Deep responses achieved after a single infusion, without induction or bridging therapy
Source: Nature
Now contrast this with phase 3 standards built around Darzalex-based regimens:
ORR: ~90–93% (MAIA, NDMM setting)
≥CR: ~30–50% range depending on backbone
MRD negativity: typically ~10–32% even in best combinations
Median PFS: ~5 years in optimal populations
Continuous multi-drug therapy required to maintain disease control
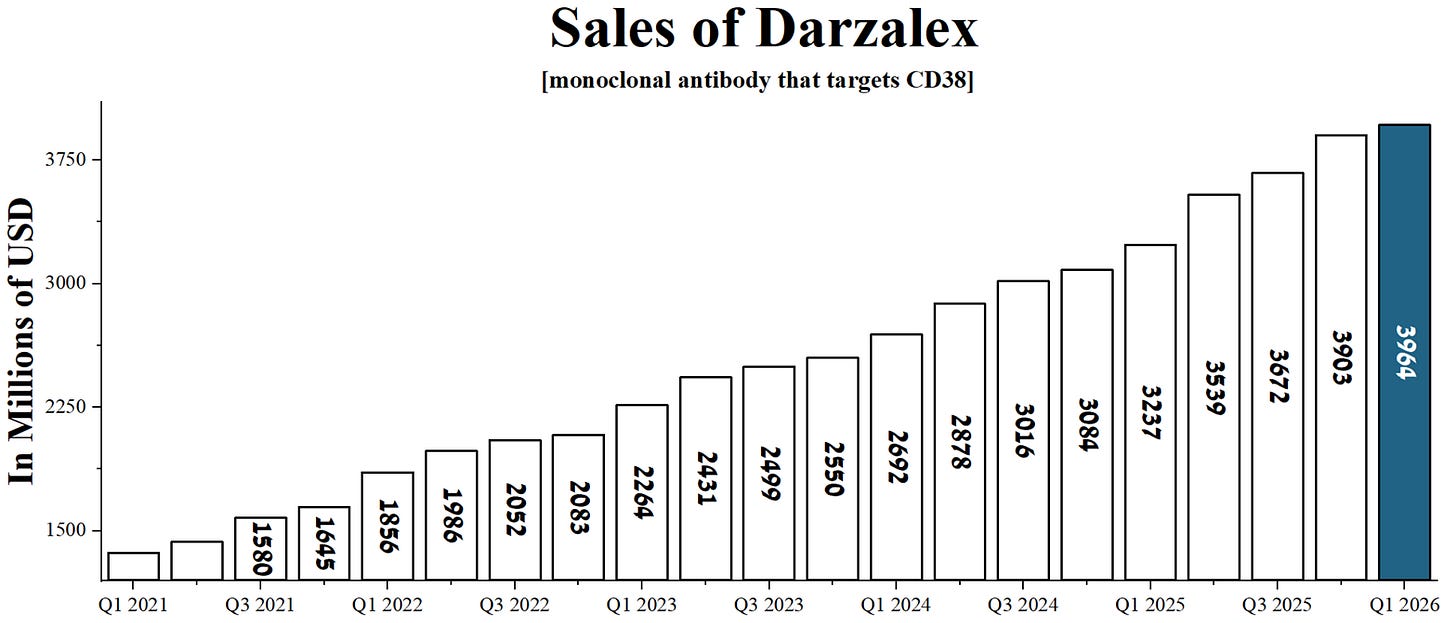
By the way, the Darzalex/Darzalex Faspro generated $3.96 billion in sales for Johnson & Johnson in Q1, up 22.5% year-over-year.
Source: graph was made by Author based on the financial reports of Johnson & Johnson
The key gap is not response rate — it is depth of response at the molecular level.
Darzalex-based regimens control disease over time.
CAR-T in this dataset is showing something different: potential complete molecular eradication in a pre-symptomatic disease window.
This is why the Dana-Farber HR-SMM dataset matters.
It suggests that when multiple myeloma is treated earlier — before full clonal expansion — CAR-T may not just extend progression-free survival, but fundamentally shift the therapeutic endpoint from chronic control to functional cure signals.
But the structural message is clear:
The gap between ~30% MRD negativity (best-in-class anti-CD38 therapy) and 100% MRD negativity (CAR-T in HR-SMM) is no longer incremental.